|
He then concluded that the rays were composed of light and negatively charged particles, a universal building block of atoms. Then Thomson compared his result with the magnetic deflection of the rays.Īs a result, Thomson could suggest that cathode rays were more than 100 times lighter than the hydrogen atom and also, he concluded that their mass was the same in whichever type of atom they came from. At first, he estimated the mass of cathode rays through the heat that was generated when the rays hit a thermal junction. He published his suggestion on 30 April 1897 following his discovery that Lenard rays could travel much further through air than expected for an atom-sized particle. To achieve this discovery, Thomson used his explorations on the properties of cathode rays. Today, the subatomic particle is known as the electron. Thomson was the first known scientist to suggest that the fundamental unit was over 1000 times smaller than an atom. 
The fact that atoms were built up from a more fundamental unit was already suggested by scientists like William Prout or Norman Lockyer. Thomson’s Cavendish Laboratory in the first years of the 1900s, as quoted “The electron: may it never be of any use to anybody!” In 1918, Thomson became Master of the Trinity College in Cambridge. Thomson himself was awarded the famous prize in 1906 “ in recognition of the great merits of his theoretical and experimental investigations on the conduction of electricity by gases.” Two years later, he was knighted. Seven of his research assistants and his son were able to win the Nobel Prizes in physics. Thomson was known to be an excellent teacher. When Thomson became Cavendish Professor of Physics, Ernest Rutherford was among his students and later on, he succeeded Thomson in the post. In 1876, he enrolled at Trinity College, Cambridge where he received his Bachelor’s and Master’s degree. Joseph John Thomson was born in 1856 in Manchester, England and was taught mainly in private schools at the beginning. Thomson, “Cathode rays” Philosophical Magazine, 44, 293 (1897). “As the cathode rays carry a charge of negative electricity, are deflected by an electrostatic force as if they were negatively electrified, and are acted on by a magnetic force in just the way in which this force would act on a negatively electrified body moving along the path of these rays, I can see no escape from the conclusion that they are charges of negative electricity carried by particles of matter.” Thomson was awarded the 1906 Nobel Prize in Physics for the discovery of the electron and for his work on the conduction of electricity in gases. These deflections could only happen if a large portion of the positive charge was located at the nucleus.On April 30, 1897, English physicist Joseph John Thomson gave the first experimental proof of the electron, which had been already theoretically predicted by Johnstone Stoney. The experiment included radon, radium, and bismuth alpha particle emitters. They utilized an alpha particle detector that produced fluorescent light spots when alpha particles struck it. Hans Geiger and Ernest Marsden conducted the experiment in 1909, directing an alpha-particle (helium nuclei) beam towards thin gold foil and then measuring the angle of Rutherford's scattered alpha particles as a consequence of these particles scattering off gold foil atoms. Rutherford's idea was to look for alpha particles with very high deflection angles, angles that were not expected from any theory of matter at the time. In 1909 Rutherford carried out the gold foil experiment in which he discovered the nuclear nature of atoms by deflecting alpha particles through a thin gold foil. JJ Thomson's plum pudding model of the atom, Tezcan - StudySmarter Originals Discovery of the electron: Ernest Rutherford and the gold foil experiment 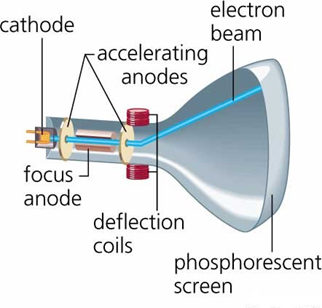
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
